Siddhartha Nanda, a Materials Science & Engineering graduate student and member of Dr. Hadi Khani's lab, has just had an article published in Energy & Environmental Science that answers the important question, "Is a non-flammable electrolyte safer?"
Potassium‑ion batteries (KIBs) are being explored as a cheaper and more sustainable alternative to today’s lithium‑ion batteries (LIBs) because potassium is abundant, works well with graphite electrodes, and relies less on scarce metals. However, the actual safety of K-ion batteries when they breakdown due to high temperatures has not been investigated thoroughly.
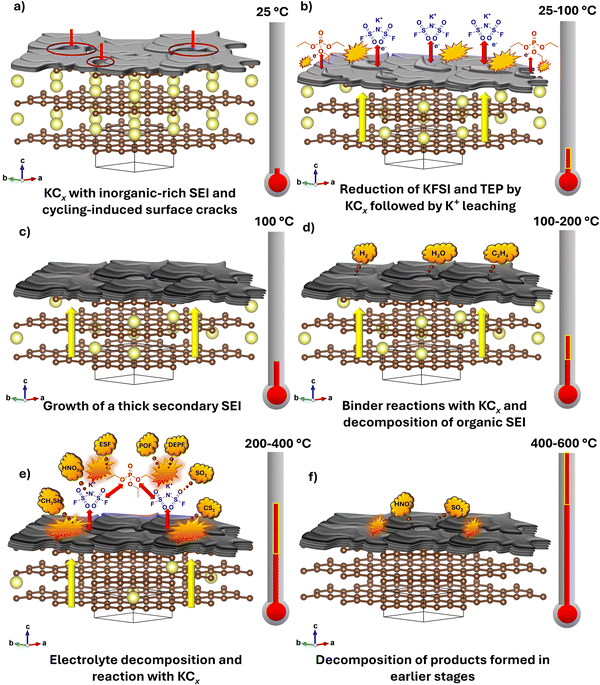
The paper, "Thermal decomposition pathways and interfacial reactivity in potassium-ion batteries: focus on the electrolyte and anode," looked at one of the "safer" K-ion battery designs that used a low-flammability liquid electrolyte. Nanda and the co-authors found that although non-flammable, this electrolyte can still undergo a chemical breakdown at high temperatures and release a large amount of heat - almost twice as much as conventional carbonate solvent based electrolytes used in LIBs, while also producing toxic and reactive byproducts. In a simplified two‑component system consisting of a fully charged graphite anode mixed with an equal mass of electrolyte, the total heat released by the Potassium system is lower but it also begins to react at lower temperatures than its lithium counterpart, which raises safety concerns that were previously not well known.
Nanda found that, overall, safety depends heavily on how the different components of a battery (anode, electrolyte, cathode, etc.) interact with each other, especially at their interfaces, and that low-flammability electrolytes do not necessarily equal a safer battery. Carefully designing interfaces - or where the materials meet - is the key to making potassium-ion batteries safer.
Other contributing authors include Andrei Dolocan, a TMI staff Scientist, Doosoo Kim, from Hadi Khani's lab, Ayrton Yanyachi and Ofodike Ezekoye from the Walker Department of Mechanical Engineering at The University of Texas, Saurabh Sandip Satpute and Kami Hull from the Department of Chemistry at UT, and Donal Finegan from the National Laboratory of the Rockies.
Read the full paper, "Thermal decomposition pathways and interfacial reactivity in potassium-ion batteries: focus on the electrolyte and anode," at Energy & Environmental Science.

